DOXORUBICIN
Doxorubicin is an anthracycline chemotherapeutic that intercalates DNA and generates free radicals. Side effects include cardiotoxicity, myelosuppression, and alopecia. Only GMP materials will be supplied, logistics all according to GDP.
About MedicaPharma
MedicaPharma distributes high-quality active pharmaceutical ingredients (APIs) to hospitals, commercial (compounding) pharmacies, research institutes, and universities worldwide.
Network of over +400 GMP API producers
Let us handle your sourcing / supply activities. Highly experienced in sourcing specialty raw pharmaceutical ingredients from niche GMP manufacturers around the world.
Why Choose MedicaPharma
Niche API specialist - Pro-active supply partner - High service level - Global network - Logistics according to GDP regulations
Product Description
Mechanism of Action
Doxorubicin is an anthracycline chemotherapeutic agent with multiple cytotoxic mechanisms. It intercalates into DNA, inhibits topoisomerase II, induces doublestrand breaks, and generates reactive oxygen species through irondependent redox cycling. It disrupts replication and transcription, leading to apoptosis in rapidly dividing cells.
Benefits and Advantages
Used in cancermodel development, DNAdamage signalling research, topoisomerase II inhibition studies, ROSgeneration assays and multidrugresistance (MDR) investigations.
Side Effects and Risks
Risks include dosedependent cardiomyopathy, myelosuppression, alopecia, mucositis, extravasation necrosis and secondary malignancies. Handle with cytotoxicanthracycline precautions.
Only GMP materials will be supplied, logistics all according to GDP.
Datasheet
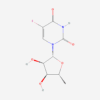
| Molecular Formula | C27H29NO11 |
|---|---|
| Molecular Weight | 543.5 g/mol |
| CAS Number | 23214-92-8 |
| Storage Condition | Commercially available doxorubicin hydrochloride lyophilized powder for injection should be stored in a dry place protected from sunlight. When stored at 15-30 °C, Adriamycin RDF or Rubex has an expiration date of 3 or 2 years, respectively, following the date of manufacture. Doxorubicin hydrochloride conventional (nonencapsulated) injection should be protected from light and refrigerated at 2-8 °C; when stored under these conditions, the injection is stable for 18 months |
| Solubility | ~10 mg/ml |
| Purity | Purity information is available upon request (COA). |
| Synonym | doxorubicin; Adriamycin; 23214-92-8; Adriablastin; Doxorubicine |
| IUPAC/Chemical Name | (7S,9S)-7-[(2R,4S,5S,6S)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy-6,9,11-trihydroxy-9-(2-hydroxyacetyl)-4-methoxy-8,10-dihydro-7H-tetracene-5,12-dione |
| InChl Key | AOJJSUZBOXZQNB-TZSSRYMLSA-N |
| InChl Code | InChI=1S/C27H29NO11/c1-10-22(31)13(28)6-17(38-10)39-15-8-27(36,16(30)9-29)7-12-19(15)26(35)21-20(24(12)33)23(32)11-4-3-5-14(37-2)18(11)25(21)34/h3-5,10,13,15,17,22,29,31,33,35-36H,6-9,28H2,1-2H3/t10-,13-,15-,17-,22+,27-/m0/s1 |
| References |
3D Conformer.
(Click, turn or enlarge)
Download our GMP API Product List.
MedicaPharma is an EU-based supplier of GMP-certified APIs that serves leading healthcare institutions and research organizations.
Click here to download our full API product list.