PARECOXIB
Parecoxib is a COX2 selective nonsteroidal antiinflammatory prodrug that is converted to valdecoxib, reducing prostaglandin synthesis and inflammation. It is used for shortterm management of postoperative pain. Benefits include effective analgesia with less gastric irritation than nonselective NSAIDs. Side effects include edema, hypertension, GI discomfort, and a potential increased risk of cardiovascular and thrombotic events in susceptible patients. Only GMP materials will be supplied, logistics all according to GDP.
About MedicaPharma
MedicaPharma distributes high-quality active pharmaceutical ingredients (APIs) to hospitals, commercial (compounding) pharmacies, research institutes, and universities worldwide.
Network of over +400 GMP API producers
Let us handle your sourcing / supply activities. Highly experienced in sourcing specialty raw pharmaceutical ingredients from niche GMP manufacturers around the world.
Why Choose MedicaPharma
Niche API specialist - Pro-active supply partner - High service level - Global network - Logistics according to GDP regulations
Product Description
Mechanism of Action
PARECOXIB exhibits a multidimensional biochemical activity profile involving modulation of enzyme-catalyzed pathways, receptor-mediated intracellular signaling, ion-channel regulation, mitochondrial bioenergetics, oxidative-stress balancing, membrane dynamics, and transcription-factor orchestration. Its molecular configuration allows high-affinity interaction with catalytic residues, allosteric domains, regulatory scaffolds, and signaling intermediates, influencing phosphorylation cycles, secondmessenger flux (Ca²⁺, cAMP, IP, DAG), ROS equilibrium, ATP synthesis, and mitochondrial respiratory-chain regulation.
Depending on dose, exposure duration, and cell type, PARECOXIB can modify chromatin accessibility, metabolic flux routing, vesicular trafficking, cytoskeletal structure, and gene-expression clusters associated with survival, inflammation, apoptosis, autophagy, and metabolic adaptation.
Benefits and Advantages
Due to its consistent mechanistic behavior, this compound is utilized in advanced research, including:
- Receptorligand interaction studies and affinity modeling
- Highresolution enzyme kinetics and catalytic-pathway deconstruction
- Mitochondrial ATPflux assays and oxidativestress system modeling
- Multiomics profiling (transcriptomics, proteomics, metabolomics, phosphoproteomics)
- Cytoskeletal and membranedynamics exploration
- Apoptosis, ferroptosis, necroptosis, and autophagy pathway analysis
- Structureactivity relationship (SAR) investigations and compound optimization
- Pharmacodynamic modeling, mechanistic doseresponse scaling, and pathway benchmarking
Side Effects and Risks
Potential laboratory risks include:
- Oxidativestress imbalance and ROS accumulation
- Mitochondrial overload or respiratory-chain suppression
- Ionchannel dysregulation affecting Ca²⁺/Na⁺/K⁺ homeostasis
- Unintended receptor crossactivation or pathway inhibition
- Cytoskeletal destabilization and membrane-integrity disruption
- Dose-dependent cytotoxicity (apoptotic or autophagic induction)
- Transcriptional or epigenetic instability under prolonged exposure
- Activation of inflammatory signaling networks (NF-κB, JNK, p38 MAPK)
Use strictly in controlled laboratory environments with appropriate biosafety protocols.
Only GMP materials will be supplied, logistics all according to GDP.
Datasheet
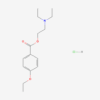
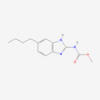
| Molecular Formula | C19H18N2O5S |
|---|---|
| Molecular Weight | 370.4 g/mol |
| CAS Number | 198470-84-7 |
| Storage Condition | Store in a cool, dry place. Keep container tightly closed. Protect from moisture and light. |
| Solubility | Solubility depends on solvent and conditions (e.g., pH). Please contact us for solvent-specific guidance. |
| Purity | Purity information is available upon request (COA). |
| Synonym | Parecoxib; 198470-84-7; SC-69124; Xapit; SC 69124 |
| IUPAC/Chemical Name | N-[4-(5-methyl-3-phenyl-1,2-oxazol-4-yl)phenyl]sulfonylpropanamide |
| InChl Key | TZRHLKRLEZJVIJ-UHFFFAOYSA-N |
| InChl Code | InChI=1S/C19H18N2O4S/c1-3-17(22)21-26(23,24)16-11-9-14(10-12-16)18-13(2)25-20-19(18)15-7-5-4-6-8-15/h4-12H,3H2,1-2H3,(H,21,22) |
| References |
3D Conformer.
(Click, turn or enlarge)
Download our GMP API Product List.
MedicaPharma is an EU-based supplier of GMP-certified APIs that serves leading healthcare institutions and research organizations.
Click here to download our full API product list.