DACOMITINIB
Daclizumab is a monoclonal antibody targeting IL2 receptors, used in transplant rejection prevention. Side effects include infections, liver dysfunction, and skin reactions. Only GMP materials will be supplied, logistics all according to GDP.
About MedicaPharma
MedicaPharma distributes high-quality active pharmaceutical ingredients (APIs) to hospitals, commercial (compounding) pharmacies, research institutes, and universities worldwide.
Network of over +400 GMP API producers
Let us handle your sourcing / supply activities. Highly experienced in sourcing specialty raw pharmaceutical ingredients from niche GMP manufacturers around the world.
Why Choose MedicaPharma
Niche API specialist - Pro-active supply partner - High service level - Global network - Logistics according to GDP regulations
Product Description
Mechanism of Action
Dacomitinib is an irreversible, panErbB tyrosinekinase inhibitor that covalently binds to EGFR, HER2, and HER4. By blocking ATP binding, it shuts down downstream MAPK/ERK and PI3K/AKT signalling, leading to reduced tumour-cell proliferation, survival, and metastasis. Unlike reversible TKIs, its covalent bond maintains prolonged pathway suppression.
Benefits and Advantages
Used in EGFRmutant cancer research, panHER pathway modelling, resistance mechanism studies (e.g., T790M, C797S), kinaseinhibitor SAR profiling, and receptordimerisation assays.
Side Effects and Risks
Risks include severe dermatologic toxicity, diarrhea, hepatotoxicity, stomatitis, interstitial lung disease and ocular irritation. Handle under advanced kinaseinhibitor cytotoxic precautions.
Only GMP materials will be supplied, logistics all according to GDP.
Datasheet
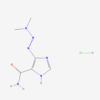
| Molecular Formula | C24H25ClFN5O2 |
|---|---|
| Molecular Weight | 469.9 g/mol |
| CAS Number | 1110813-31-4 |
| Storage Condition | Store in a cool, dry place. Keep container tightly closed. Protect from moisture and light. |
| Solubility | <1 mg/mL |
| Purity | Purity information is available upon request (COA). |
| Synonym | Dacomitinib; 1110813-31-4; PF-00299804; Dacomitinib anhydrous; Vizimpro |
| IUPAC/Chemical Name | (E)-N-[4-(3-chloro-4-fluoroanilino)-7-methoxyquinazolin-6-yl]-4-piperidin-1-ylbut-2-enamide |
| InChl Key | LVXJQMNHJWSHET-AATRIKPKSA-N |
| InChl Code | InChI=1S/C24H25ClFN5O2/c1-33-22-14-20-17(24(28-15-27-20)29-16-7-8-19(26)18(25)12-16)13-21(22)30-23(32)6-5-11-31-9-3-2-4-10-31/h5-8,12-15H,2-4,9-11H2,1H3,(H,30,32)(H,27,28,29)/b6-5+ |
| References |
3D Conformer.
(Click, turn or enlarge)
Download our GMP API Product List.
MedicaPharma is an EU-based supplier of GMP-certified APIs that serves leading healthcare institutions and research organizations.
Click here to download our full API product list.