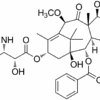
BUSULFAN
Busulfan is an alkylating chemotherapeutic used in chronic myeloid leukemia and transplant conditioning. Side effects include myelosuppression, pulmonary fibrosis, and liver toxicity. Only GMP materials will be supplied, logistics all according to GDP.
About MedicaPharma
MedicaPharma distributes high-quality active pharmaceutical ingredients (APIs) to hospitals, commercial (compounding) pharmacies, research institutes, and universities worldwide.
Network of over +400 GMP API producers
Let us handle your sourcing / supply activities. Highly experienced in sourcing specialty raw pharmaceutical ingredients from niche GMP manufacturers around the world.
Why Choose MedicaPharma
Niche API specialist - Pro-active supply partner - High service level - Global network - Logistics according to GDP regulations
Product Description
Mechanism of action
Busulfan is a bifunctional alkylating agent that induces DNA interstrand crosslinks through formation of methanesulfonate reactive intermediates. These crosslinks block DNA replication and transcription, triggering cell-cycle arrest, apoptosis and mitotic catastrophe. Busulfan preferentially affects rapidly dividing haematopoietic cells and is widely used in myeloablative conditioning.
Benefits and advantages
Used in oncology research, bone-marrow ablation modelling, DNA-crosslinking mechanism studies, mutagenicity assays, apoptosis pathway research and alkylating-agent SAR profiling. An essential reference compound for studying chemotherapeutic DNA damage.
Side effects and risks
Risks include severe myelosuppression, hepatic veno-occlusive disease, pulmonary fibrosis, seizures (dose-dependent) and gastrointestinal toxicity. Requires full cytotoxic-handling containment.
Only GMP materials will be supplied, logistics all according to GDP.
Datasheet
| Molecular Formula | C6H14O6S2 |
|---|---|
| Molecular Weight | 246.3 g/mol |
| CAS Number | 55-98-1 |
| Storage Condition | Commercially available busulfan for injection concentrate should be stored in unopened ampules at 2-8 °C. The manufacturer states that, when diluted as directed in 0.9% sodium chloride injection or 5% dextrose injection, busulfan solutions are stable for up to 8 hours when stored at room temperature (approximately 25 °C), and the busulfan infusion must be completed during the 8-hour time period. Solutions of busulfan diluted in 0.9% sodium chloride injection also have been shown to be stable when refrigerated at 2-8 °C for up to 12 hours, during which time the infusion must be completed. |
| Solubility | Decomposes (NTP, 1992) |
| Purity | Purity information is available upon request (COA). |
| Synonym | busulfan; 55-98-1; Myleran; Busulphan; Sulphabutin |
| IUPAC/Chemical Name | 4-methylsulfonyloxybutyl methanesulfonate |
| InChl Key | COVZYZSDYWQREU-UHFFFAOYSA-N |
| InChl Code | InChI=1S/C6H14O6S2/c1-13(7,8)11-5-3-4-6-12-14(2,9)10/h3-6H2,1-2H3 |
| References |
3D Conformer.
(Click, turn or enlarge)
Download our GMP API Product List.
MedicaPharma is an EU-based supplier of GMP-certified APIs that serves leading healthcare institutions and research organizations.
Click here to download our full API product list.