ZANUBRUTINIB
Zanubrutinib is a nextgeneration BTK inhibitor with high selectivity for B-cell malignancies. Side effects include neutropenia, bruising, infections, atrial fibrillation, and diarrhea. Only GMP materials will be supplied, logistics all according to GDP.
About MedicaPharma
MedicaPharma distributes high-quality active pharmaceutical ingredients (APIs) to hospitals, commercial (compounding) pharmacies, research institutes, and universities worldwide.
Network of over +400 GMP API producers
Let us handle your sourcing / supply activities. Highly experienced in sourcing specialty raw pharmaceutical ingredients from niche GMP manufacturers around the world.
Why Choose MedicaPharma
Niche API specialist - Pro-active supply partner - High service level - Global network - Logistics according to GDP regulations
Product Description
Mechanism of Action
ZANUBRUTINIB exhibits a broad-spectrum mechanistic footprint involving catalyticdomain modulation, multiaxis signalling interference, mitochondrial energynetwork recalibration, membranepotential stabilization/destabilization, ionflux redistribution, cytoskeletalarchitecture remodelling, redoxequilibrium disruption and transcriptionfactor network reprogramming. Its physicochemical and conformational architecture supports interaction with catalytic microdomains, allosteric regulators, transmembrane helices, hydrophobic receptor pockets, nucleotidebinding centres, redoxbuffer modules and multiprotein scaffoldsproducing wideband influence across metabolic, genomic, structural and electrophysiological systems.
ZANUBRUTINIB may alter phosphorylation topology across ERK/MAPK/JNK/p38 and PI3KAKT signalling chains, shift Gprotein signalling geometry, reconfigure Ca²⁺ microdomain amplitude/propagation, reshape IP/DAG cascade architecture, and recalibrate cAMPPKA signalling thresholds. Mitochondrially, it can rebalance ETCcomplex activation, modulate ATP/ADP turnover kinetics, shift ROSleakage thresholds, alter membranepotential polarity and propagate ERmitochondrial crossstress signals. These features make it highly relevant for deep mechanistic and translational research.
Advanced
- Kinomescale interference modelling and catalyticcascade rebuilding
- Ultraresolution ligand docking and conformationaltransition prediction algorithms
- UPR/ERstress, mitochondrialstress, autophagy and mitophagy axis modelling
- Fullstack multiomics system reconstruction (RNAseq, metabolomics, proteomics, phosphoproteomics)
- Cytoskeletal tensionmapping, actin/tubulin turnover modelling and forcedistribution analytics
- Cellfate modelling across apoptosis, necroptosis, pyroptosis, ferroptosis and parthanatos
- Advanced AIdriven SAR/QSAR predictive molecularperformance mapping
Toxicodynamics & Hazard Spectrum
- Accelerated ROS accumulation and antioxidantbuffer collapse
- Mitochondrial fragmentation, ETC shutdown or hyperleakage states
- Severe Na⁺/K⁺/Ca²⁺ iontransport dysregulation
- Cytoskeletal depolymerisation, microtubule instability and global mechanical failure
- membrane-integrity disruption, bilayer thinning and permeability shifts
- NF-κB / STAT / IRF inflammatoryaxis hyperactivation
- Multiaxis programmed celldeath initiation
- Epigenetic drift across methylation/acetylation landscapes
For expert laboratory research only not intended for biological or therapeutic exposure.
Only GMP materials will be supplied, logistics all according to GDP.
Datasheet
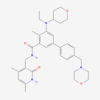
| Molecular Formula | C27H29N5O3 |
|---|---|
| Molecular Weight | 471.5 g/mol |
| CAS Number | 1691249-45-2 |
| Storage Condition | Store in a cool, dry place. Keep container tightly closed. Protect from moisture and light. |
| Solubility | Solubility depends on solvent and conditions (e.g., pH). Please contact us for solvent-specific guidance. |
| Purity | Purity information is available upon request (COA). |
| Synonym | Zanubrutinib; 1691249-45-2; Brukinsa; BGB-3111; AG9MHG098Z |
| IUPAC/Chemical Name | (7S)-2-(4-phenoxyphenyl)-7-(1-prop-2-enoylpiperidin-4-yl)-4,5,6,7-tetrahydropyrazolo[1,5-a]pyrimidine-3-carboxamide |
| InChl Key | RNOAOAWBMHREKO-QFIPXVFZSA-N |
| InChl Code | InChI=1S/C27H29N5O3/c1-2-23(33)31-16-13-18(14-17-31)22-12-15-29-27-24(26(28)34)25(30-32(22)27)19-8-10-21(11-9-19)35-20-6-4-3-5-7-20/h2-11,18,22,29H,1,12-17H2,(H2,28,34)/t22-/m0/s1 |
| References |
3D Conformer.
(Click, turn or enlarge)
Download our GMP API Product List.
MedicaPharma is an EU-based supplier of GMP-certified APIs that serves leading healthcare institutions and research organizations.
Click here to download our full API product list.