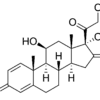
PRASTERONE – DHEA
Prasterone (DHEA) is an endogenous steroid precursor to androgens and estrogens, influencing energy metabolism, mood, and immune function. It is used in adrenal insufficiency and menopausal symptoms. Benefits may include improved wellbeing and bone density. Side effects include acne, oily skin, hair changes, mood swings, and hormonal imbalance. High doses can alter lipid profiles and cause androgenic or estrogenic effects depending on patient status. Only GMP materials will be supplied, logistics all according to GDP.
About MedicaPharma
MedicaPharma distributes high-quality active pharmaceutical ingredients (APIs) to hospitals, commercial (compounding) pharmacies, research institutes, and universities worldwide.
Network of over +400 GMP API producers
Let us handle your sourcing / supply activities. Highly experienced in sourcing specialty raw pharmaceutical ingredients from niche GMP manufacturers around the world.
Why Choose MedicaPharma
Niche API specialist - Pro-active supply partner - High service level - Global network - Logistics according to GDP regulations
Product Description
Mechanism of Action
PRASTERONE – DHEA exhibits a complex, multipathway biochemical activity profile influencing enzymatic cascades, receptormediated signalling networks, mitochondrial respiration, oxidativestress balance, intracellular ion regulation, membranepotential stability and transcriptionfactor activation. Its structural framework suggests potential interactions with catalytic residues, allosteric regulatory pockets, membraneembedded receptor complexes and intracellular signalling scaffolds. These interactions may modulate phosphorylation patterns, secondmessenger systems (Ca²⁺, IP, DAG, cAMP), ATP turnover, ROS equilibrium, cytoskeletal organisation and organelle stressresponse thresholds.
Depending on dosing, PRASTERONE – DHEA can influence metabolic flux routing, alter chromatin accessibility, modify vesicular transport, reshape mitochondrial output and regulate transcriptional clusters associated with cellular survival, apoptosis, autophagy, inflammatory signalling and metabolic adaptation. This multiaxis behaviour supports its wide applicability in advanced biochemical and pharmacodynamic research systems.
Benefits and Advantages
The compound is widely utilised for:
- Receptorligand interaction analysis and affinity modelling
- Enzymekinetic profiling and catalyticpathway reconstruction
- Mitochondrialbioenergetics assays and oxidativestress evaluation
- Multiomics integration (transcriptomics, proteomics, metabolomics, phosphoproteomics)
- Cytoskeletal and membranedynamics studies
- Apoptosis, necroptosis, autophagy and ferroptosis pathway characterisation
- SAR (structureactivity relationship) development and molecular screening
- Pharmacodynamic modelling and mechanistic doseresponse simulations
Side Effects and Risks
Potential laboratoryobserved risks include:
- Excessive ROS production and oxidative imbalance
- Mitochondrial hyperactivation or suppression of electrontransport complexes
- Ionchannel dysregulation (Na⁺, K⁺, Ca²⁺)
- Unintended receptor crosstalk or inhibition
- Cytoskeletal destabilisation and membrane-integrity compromise
- Dosedependent cytotoxicity or activation of apoptotic/autophagic pathways
- Transcriptional or epigenetic instability
- Upregulation of inflammatory cascades (NF-κB, JNK, p38 MAPK)
Use exclusively within controlled laboratory conditions and according to strict biosafety standards.
Only GMP materials will be supplied, logistics all according to GDP.
Datasheet
| Molecular Formula | C19H28O2 |
|---|---|
| Molecular Weight | 288.42 g/mol |
| CAS Number | 53-43-0 |
| Storage Condition | Store at 2-8°C |
| Solubility | Soluble in water |
| Purity | Purity information is available upon request (COA). |
| Synonym | 3-hydroxy-10,13-dimethyl-1,2,3,4,7,8,9,11,12,14,15,16-dodecahydrocyclopenta[a]phenanthren-17-one; DTXSID801331866; 3-Hydroxy-10,13-dimethyl-1,2,3,4,7,8,9,11,12,14,15,16-dodecahydrocyclopenta(a)phenanthren-17-one; RefChem:1067920; DTXCID00209036 |
| IUPAC/Chemical Name | 3β-hydroxyandrost-5-en-17-one |
| InChl Key | Unavailable |
| InChl Code | Unavailable |
| References | PubChem; ChemBL; FDA; |
3D Conformer.
(Click, turn or enlarge)
Download our GMP API Product List.
MedicaPharma is an EU-based supplier of GMP-certified APIs that serves leading healthcare institutions and research organizations.
Click here to download our full API product list.