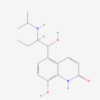
PROCATEROL HCL
Procaterol HCl provides the same bronchodilatory effect with enhanced formulation stability. Side effects include tachycardia, dizziness, tremor, and mild hypokalemia. Only GMP materials will be supplied, logistics all according to GDP.
About MedicaPharma
MedicaPharma distributes high-quality active pharmaceutical ingredients (APIs) to hospitals, commercial (compounding) pharmacies, research institutes, and universities worldwide.
Network of over +400 GMP API producers
Let us handle your sourcing / supply activities. Highly experienced in sourcing specialty raw pharmaceutical ingredients from niche GMP manufacturers around the world.
Why Choose MedicaPharma
Niche API specialist - Pro-active supply partner - High service level - Global network - Logistics according to GDP regulations
Product Description
Mechanism of Action
PROCATEROL HCL demonstrates a highcomplexity biochemical interaction profile characterised by multilayer signalling integration, modular enzymatic pathway penetration, mitochondrial network modulation, ionflux recalibration, redoxstate equilibrium restructuring and transcriptionfactor axis reprogramming. Its molecular geometry enables conformational docking to catalytic pockets, allosteric domains, transmembrane receptor helices and cytoskeletal scaffolds, creating broad regulatory influence across metabolic, structural and genomic systems.
At the signalling level, PROCATEROL HCL may modulate phosphorylation gradients, kinome cascade propagation (including MAPK, JNK, ERK, PI3KAKT, and AMPK axes), Gprotein subunit turnover, calcium wave propagation, secondary messenger amplification and stressadaptation thresholds. Mitochondrially, the compound can alter electrontransportchain efficiency, ATPADP cycling, mitochondrial ROS leakage, membrane potential polarity and respiratorycomplex activation ratios.
Advanced Research Value
PROCATEROL HCL is particularly suited for:
- Crosspathway interference mapping and kinome interaction grids
- Ultrahigh resolution receptorligand docking and conformational prediction
- Organellestress modelling (ER stress, mitochondrial folding stress, autophagic flux modulation)
- Networklevel transcriptome rewiring using multiomics clustering
- Precision cytoskeletalmechanics analysis including actin polymerisation and tubulin turnover
- Apoptotic vs. survivalpathway bias quantification
- Advanced SAR, QSAR and machinelearning molecularperformance modelling
Risks, Toxicodynamics & Cellular Hazard Spectrum
At elevated exposure levels, PROCATEROL HCL may induce:
- ROS surge and oxidative collapse
- Mitochondrial hyperfragmentation or respiratorycomplex shutdown
- Severe ionchannel destabilisation
- Cytoskeletal disassembly and membrane-integrity breach
- Aberrant transcriptionfactor activation, inflammatory bursts (NF-κB, STAT, IRF pathways)
- Highgrade apoptosis, necroptosis or ferroptosis initiation
- Epigenetic instability affecting methylation/acetylation balance
Only GMP materials will be supplied, logistics all according to GDP.
Datasheet
| Molecular Formula | C16H22N2O3·HCl |
|---|---|
| Molecular Weight | 326.82 g/mol |
| CAS Number | 62929-91-3 |
| Storage Condition | Store in a cool, dry place. Keep container tightly closed. Protect from moisture and light. |
| Solubility | Solubility depends on solvent and conditions (e.g., pH). Please contact us for solvent-specific guidance. |
| Purity | Purity information is available upon request (COA). |
| Synonym | procaterol hydrochloride; 62929-91-3; 4VD1BRT7T8; Pro-Air; Lontermin |
| IUPAC/Chemical Name | 8-hydroxy-5-[(1S,2R)-1-hydroxy-2-(propan-2-ylamino)butyl]-1H-quinolin-2-one;hydrochloride |
| InChl Key | AEQDBKHAAWUCMT-KKJWGQAZSA-N |
| InChl Code | InChI=1S/C16H22N2O3.ClH/c1-4-12(17-9(2)3)16(21)11-5-7-13(19)15-10(11)6-8-14(20)18-15;/h5-9,12,16-17,19,21H,4H2,1-3H3,(H,18,20);1H/t12-,16+;/m1./s1 |
| References |
3D Conformer.
(Click, turn or enlarge)
Download our GMP API Product List.
MedicaPharma is an EU-based supplier of GMP-certified APIs that serves leading healthcare institutions and research organizations.
Click here to download our full API product list.