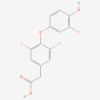
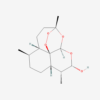
Molnupiravir
Molnupiravir is an orally active prodrug of NHC/EIDD-1931 used in antiviral pharmaceutical research. After conversion to its active triphosphate form, it can be incorporated into viral RNA and promote replication errors in RNA-virus models. Potential risks include gastrointestinal effects, dizziness, embryo-fetal concerns and genotoxicity-related questions. Only GMP materials will be supplied, logistics all according to GDP.
About MedicaPharma
MedicaPharma distributes high-quality active pharmaceutical ingredients (APIs) to hospitals, commercial (compounding) pharmacies, research institutes, and universities worldwide.
Network of over +400 GMP API producers
Let us handle your sourcing / supply activities. Highly experienced in sourcing specialty raw pharmaceutical ingredients from niche GMP manufacturers around the world.
Why Choose MedicaPharma
Niche API specialist - Pro-active supply partner - High service level - Global network - Logistics according to GDP regulations
Product Description
Mechanism of Action
Molnupiravir is an orally active prodrug of beta-D-N4-hydroxycytidine, also known as NHC or EIDD-1931. After metabolic conversion and phosphorylation, the active ribonucleoside triphosphate can be incorporated into viral RNA and promote error accumulation during replication by RNA-dependent RNA polymerases.
Benefits and Advantages
Molnupiravir is used in antiviral pharmaceutical research, prodrug activation studies, RNA-virus replication models and comparative work on nucleoside analogue strategies. It is relevant for evaluating metabolic conversion, formulation stability and polymerase-driven mutagenesis mechanisms.
Side Effects and Risks
Potential risks include gastrointestinal effects, dizziness, cytotoxicity concerns, embryo-fetal risk considerations and genotoxicity-related questions that require careful assessment. Handling and use must remain within controlled pharmaceutical, toxicological and regulatory environments.
Professional use only. Supplied exclusively for qualified pharmaceutical, analytical or laboratory environments.
Only GMP materials will be supplied, logistics all according to GDP.
Datasheet
| Molecular Formula | C13H19N3O7 |
|---|---|
| Molecular Weight | 329.31 g/mol |
| CAS Number | 2349386-89-4, 2492423-29-5 |
| Storage Condition | Store in a cool, dry place. Keep container tightly closed. Protect from moisture and light. |
| Solubility | Soluble in DMSO; solubility depends on solvent and conditions. |
| Purity | Purity information is available upon request (COA). |
| Synonym | Molnupiravir; EIDD-2801; MK-4482; Lagevrio; N-hydroxy-5-O-isobutyrylcytidine |
| IUPAC/Chemical Name | [(2R,3S,4R,5R)-3,4-dihydroxy-5-[4-(hydroxyamino)-2-oxopyrimidin-1-yl]oxolan-2-yl]methyl 2-methylpropanoate |
| InChl Key | HTNPEHXGEKVIHG-QCNRFFRDSA-N |
| InChl Code | InChI=1S/C13H19N3O7/c1-6(2)12(20)22-5-8-9(17)10(18)11(23-8)16-4-3-7(14-21)15-13(16)19/h3-4,6,8-11,17-18,21H,5H2,1-2H3/t8-,9-,10+,11-/m1/s1 |
| References | https://pubchem.ncbi.nlm.nih.gov/compound/145996610; small-molecule |
3D Conformer.
(Click, turn or enlarge)
Download our GMP API Product List.
MedicaPharma is an EU-based supplier of GMP-certified APIs that serves leading healthcare institutions and research organizations.
Click here to download our full API product list.