METOCLOPRAMIDE DIHYDROCHLORIDE
Metoclopramide dihydrochloride provides identical prokinetic and antiemetic effects with enhanced solubility. Side effects include fatigue, restlessness, and movement disorders. Only GMP materials will be supplied, logistics all according to GDP.
About MedicaPharma
MedicaPharma distributes high-quality active pharmaceutical ingredients (APIs) to hospitals, commercial (compounding) pharmacies, research institutes, and universities worldwide.
Network of over +400 GMP API producers
Let us handle your sourcing / supply activities. Highly experienced in sourcing specialty raw pharmaceutical ingredients from niche GMP manufacturers around the world.
Why Choose MedicaPharma
Niche API specialist - Pro-active supply partner - High service level - Global network - Logistics according to GDP regulations
Product Description
Mechanism of Action
METOCLOPRAMIDE DIHYDROCHLORIDE demonstrates an extensive, multiaxis biochemical activity profile involving coordinated modulation of receptormediated signalling, enzymeregulated catalytic cascades, mitochondrial bioenergetics, intracellular ion regulation, and transcriptomic pathway dynamics. Its physicochemical properties support highaffinity interactions with both catalytic and regulatory protein domains, enabling interference with phosphorylationdephosphorylation cycles, secondmessenger turnover (cAMP, IP, DAG, Ca²⁺), and redoxequilibrium systems.
In cellular environments, METOCLOPRAMIDE DIHYDROCHLORIDE may influence mitochondrial membrane potential, ATP synthesis efficiency, ROS generation and antioxidant neutralisation capacity. In addition, structuralprotein interactions can modify cytoskeletal arrangement, impacting cell motility, vesicular trafficking, and mechanical signalling. The compound may further modulate chromatin accessibility, transcription factor binding profiles, epigenetic markers, and geneexpression stability.
Benefits and Advantages
Because of its reproducible biochemical footprint, METOCLOPRAMIDE DIHYDROCHLORIDE is an important tool across advanced research domains, including:
- Detailed receptorligand binding analysis and structural docking validation
- Highdefinition enzyme kinetics and catalyticdomain pathway mapping
- Mitochondrial respiration modelling, ATPflux analysis and oxidativestress pathway evaluation
- Transcriptomic, proteomic and metabolomic integration studies for fullpathway profiling
- Apoptosis, necroptosis and autophagyrelated signalling research
- Cytoskeletaldynamics studies involving actin, tubulin and intermediate filaments
- Highresolution SAR (structureactivity relationship) investigations for molecular optimisation
- Pharmacodynamic modelling for doseresponse, thresholdactivation and mechanistic benchmarking
Side Effects and Risks
Potential risks include:
- Redox imbalance and excessive ROS accumulation
- Mitochondrial overload or respiratorychain suppression
- Transcriptional pathway disruption or epigenetic instability
- Interference with ionchannel behaviour and calciumhandling regulation
- Cytoskeletal destabilisation and impaired membrane integrity
- Dosedependent cytotoxicity, apoptosis or autophagy induction
- Activation of inflammatory signalling cascades such as NF-κB, JNK, or p38
Extended exposure or elevated dosing may trigger metabolic rewiring, organelle stress responses, or longterm alterations in cellular signalling architecture. Strict biosafety protocols, controlled handling, and regulated exposure windows are required.
Only GMP materials will be supplied, logistics all according to GDP.
Datasheet
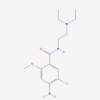
| Molecular Formula | C14H22Cl2N3O2 |
|---|---|
| Molecular Weight | 372.7 g/mol |
| CAS Number | 2576-84-3 |
| Storage Condition | Metoclopramide hydrochloride tablets, injection, and oral solution should be stored at controlled room temperature between 20-25 °C. Metoclopramide hydrochloride tablets and metoclopramide hydrochloride oral solution should be stored in tight, light-resistant containers (ie, amber glass bottles). |
| Solubility | Crystal; decomposes at 145 °C. Solubility at 25 °C (g/100 mL): water 48; ethanol (95%) 9; absolute ethanol 6; benzene 0.10; chloroform 0.10. Stable in acidic solutions. Unstable in strongly alkaline solutions. /Metoclopramide Dihydrochloride monohydrate/ |
| Purity | Purity information is available upon request (COA). |
| Synonym | Metoclopramide dihydrochloride; 2576-84-3; 5YNA80E9TO; 4-amino-5-chloro-N-[2-(diethylamino)ethyl]-2-methoxybenzamide dihydrochloride; 4-Amino-5-chloro-N-(2-(diethylamino)ethyl)-2-methoxybenzamide dihydrochloride |
| IUPAC/Chemical Name | 4-amino-5-chloro-N-[2-(diethylamino)ethyl]-2-methoxybenzamide;dihydrochloride |
| InChl Key | HPPDSFRJRMUHSC-UHFFFAOYSA-N |
| InChl Code | InChI=1S/C14H22ClN3O2.2ClH/c1-4-18(5-2)7-6-17-14(19)10-8-11(15)12(16)9-13(10)20-3;;/h8-9H,4-7,16H2,1-3H3,(H,17,19);2*1H |
| References |
3D Conformer.
(Click, turn or enlarge)
Download our GMP API Product List.
MedicaPharma is an EU-based supplier of GMP-certified APIs that serves leading healthcare institutions and research organizations.
Click here to download our full API product list.