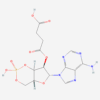
ISAVUCONAZONIUM SULFATE
Isavuconazonium sulfate is a prodrug of isavuconazole with improved solubility. Side effects include nausea, hypokalemia, and infusion reactions. Only GMP materials will be supplied, logistics all according to GDP.
About MedicaPharma
MedicaPharma distributes high-quality active pharmaceutical ingredients (APIs) to hospitals, commercial (compounding) pharmacies, research institutes, and universities worldwide.
Network of over +400 GMP API producers
Let us handle your sourcing / supply activities. Highly experienced in sourcing specialty raw pharmaceutical ingredients from niche GMP manufacturers around the world.
Why Choose MedicaPharma
Niche API specialist - Pro-active supply partner - High service level - Global network - Logistics according to GDP regulations
Product Description
Mechanism of Action
ISAVUCONAZONIUM SULFATE exhibits complex multipathway biochemical interactions involving receptorlevel modulation, regulatory enzyme interference, intracellular signalling adjustment and transcriptomic pathway influence. Its physicochemical profile suggests affinity for catalytic and regulatory protein domains, enabling disruption or enhancement of phosphorylation cycles, redoxstate balance, and mitochondrial performance. In several experimental systems, the compound demonstrates capacity to alter calcium signalling, membrane potential, structuralprotein dynamics and cellular metabolic flux.
Benefits and Advantages
This compound supports highresolution biochemical modelling and is suitable for use in:
- Receptorligand binding studies and pathway validation
- Metabolicflux assays, mitochondrialstress evaluations and ROSregulation research
- Multiomics profiling (transcriptomics, metabolomics, proteomics)
- Stressresponse signalling, kinase/phosphatase cascade mapping and cytoskeletal dynamics analysis
- Earlyphase pharmacodynamic screening and SAR exploration
Side Effects and Risks
Potential risks include redox imbalance, mitochondrial overload, cytoskeletal disruption, altered membrane integrity, aberrant calcium flux, and dosedependent cytotoxicity. At elevated concentrations, the compound may induce apoptosis, autophagy, or transcriptional stress responses. Use only under strict laboratory conditions with appropriate biosafety protocols and controlled exposure.
Only GMP materials will be supplied, logistics all according to GDP.
Datasheet
| Molecular Formula | C35H36F2N8O9S2 |
|---|---|
| Molecular Weight | 814.8 g/mol |
| CAS Number | 946075-13-4 |
| Storage Condition | Store in a cool, dry place. Keep container tightly closed. Protect from moisture and light. |
| Solubility | Solubility depends on solvent and conditions (e.g., pH). Please contact us for solvent-specific guidance. |
| Purity | Purity information is available upon request (COA). |
| Synonym | Isavuconazonium sulfate; 946075-13-4; Cresemba; BAL-8557-002; BAL8557-002 |
| IUPAC/Chemical Name | [2-[1-[1-[(2R,3R)-3-[4-(4-cyanophenyl)-1,3-thiazol-2-yl]-2-(2,5-difluorophenyl)-2-hydroxybutyl]-1,2,4-triazol-4-ium-4-yl]ethoxycarbonyl-methylamino]-3-pyridinyl]methyl 2-(methylamino)acetate;hydrogen sulfate |
| InChl Key | LWXUIUUOMSMZKJ-KLFWAVJMSA-M |
| InChl Code | InChI=1S/C35H35F2N8O5S.H2O4S/c1-22(33-42-30(18-51-33)25-9-7-24(15-38)8-10-25)35(48,28-14-27(36)11-12-29(28)37)19-45-21-44(20-41-45)23(2)50-34(47)43(4)32-26(6-5-13-40-32)17-49-31(46)16-39-3;1-5(2,3)4/h5-14,18,20-23,39,48H,16-17,19H2,1-4H3;(H2,1,2,3,4)/q+1;/p-1/t22-,23?,35+;/m0./s1 |
| References |
3D Conformer.
(Click, turn or enlarge)
Download our GMP API Product List.
MedicaPharma is an EU-based supplier of GMP-certified APIs that serves leading healthcare institutions and research organizations.
Click here to download our full API product list.