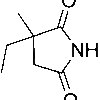
ETHIONAMIDE
Ethionamide is a second-line antitubercular drug inhibiting mycolic acid synthesis in Mycobacterium tuberculosis. Side effects include hepatotoxicity, GI upset, and neurological symptoms such as peripheral neuropathy. Only GMP materials will be supplied, logistics all according to GDP.
About MedicaPharma
MedicaPharma distributes high-quality active pharmaceutical ingredients (APIs) to hospitals, commercial (compounding) pharmacies, research institutes, and universities worldwide.
Network of over +400 GMP API producers
Let us handle your sourcing / supply activities. Highly experienced in sourcing specialty raw pharmaceutical ingredients from niche GMP manufacturers around the world.
Why Choose MedicaPharma
Niche API specialist - Pro-active supply partner - High service level - Global network - Logistics according to GDP regulations
Product Description
Mechanism of Action
Ethionamide is a prodrug activated by mycobacterial monooxygenase (EtaA). The activated form inhibits InhA, an enoylACP reductase required for mycolicacid synthesis, thereby disrupting the integrity of the mycobacterial cell wall.
Benefits and Advantages
Used in TBpathway research, prodrugactivation assays, mycolicacid biosynthesis studies, and multidrugresistance modelling.
Side Effects and Risks
Risks include hepatotoxicity, GI intolerance, neurotoxicity and hypothyroidism. Handle with antimycobacterialthioamide precautions.
Only GMP materials will be supplied, logistics all according to GDP.
Datasheet
| Molecular Formula | C8H10N2S |
|---|---|
| Molecular Weight | 166.25 g/mol |
| CAS Number | 536-33-4 |
| Storage Condition | Ethionamide film-coated tablets should be stored in tight containers at 20-25 °C. |
| Solubility | less than 1 mg/mL at 70 °F (NTP, 1992) |
| Purity | Purity information is available upon request (COA). |
| Synonym | ethionamide; 536-33-4; 2-ethylpyridine-4-carbothioamide; Trecator; Ethioniamide |
| IUPAC/Chemical Name | 2-ethylpyridine-4-carbothioamide |
| InChl Key | AEOCXXJPGCBFJA-UHFFFAOYSA-N |
| InChl Code | InChI=1S/C8H10N2S/c1-2-7-5-6(8(9)11)3-4-10-7/h3-5H,2H2,1H3,(H2,9,11) |
| References |
3D Conformer.
(Click, turn or enlarge)
Download our GMP API Product List.
MedicaPharma is an EU-based supplier of GMP-certified APIs that serves leading healthcare institutions and research organizations.
Click here to download our full API product list.