RIVASTIGMINE TARTRATE
Rivastigmine tartrate is an orally administered cholinesterase inhibitor used to treat mild to moderate dementia. It increases cholinergic neurotransmission. Side effects include GI upset, anorexia, dizziness, sweating, insomnia, and risk of bradyarrhythmias in sensitive individuals. Only GMP materials will be supplied, logistics all according to GDP.
About MedicaPharma
MedicaPharma distributes high-quality active pharmaceutical ingredients (APIs) to hospitals, commercial (compounding) pharmacies, research institutes, and universities worldwide.
Network of over +400 GMP API producers
Let us handle your sourcing / supply activities. Highly experienced in sourcing specialty raw pharmaceutical ingredients from niche GMP manufacturers around the world.
Why Choose MedicaPharma
Niche API specialist - Pro-active supply partner - High service level - Global network - Logistics according to GDP regulations
Product Description
Mechanism of Action
RIVASTIGMINE TARTRATE engages in multilayer biochemical interactions affecting enzyme cascades, receptorregulated signalling, oxidativestress systems, mitochondrial respiration, ionchannel dynamics, cytoskeletal mechanics and transcriptionfactor pathway coordination. Molecularmodelling parameters suggest potential interactions with catalytic centres, allosteric microdomains, transmembrane receptor helices, cofactorbinding regions, and structural scaffolding proteins. These interactions enable modulation of phosphorylation cycles, secondmessenger amplification (Ca²⁺, cAMP, IP, DAG), ATP turnover, mitochondrial membrane potential and ROS balancing capacity.
Benefits and Applications
- Receptorligand affinity mapping and molecular docking
- Enzymekinetic pathway modelling and catalyticflux analysis
- Mitochondrial bioenergetics profiling and redoxstate research
- Multiomics studies (transcriptomics, metabolomics, proteomics)
- Cytoskeletal architecture analysis and membranedynamics modelling
- Apoptosis, necroptosis, autophagy and ferroptosis pathway assays
- SAR and QSARdriven molecular optimisation
- Pharmacodynamic and mechanistic doseresponse modelling
Risks & Side Effects
- Oxidative imbalance and ROS escalation
- Mitochondrial overload or suppression of respiratory complexes
- Ionchannel destabilisation (Na⁺/K⁺/Ca²⁺)
- Unintended receptor crosstalk
- Cytoskeletal destabilisation
- Dosedependent cytotoxicity or programmed-cell-death activation
- Inflammatory transcriptional activation (NF-κB, JNK, MAPK)
Only GMP materials will be supplied, logistics all according to GDP.
Datasheet
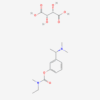
| Molecular Formula | C14H22N2O2·C4H6O6 |
|---|---|
| Molecular Weight | 400.4 g/mol |
| CAS Number | 129101-54-8 |
| Storage Condition | Store in a cool, dry place. Keep container tightly closed. Protect from moisture and light. |
| Solubility | Solubility depends on solvent and conditions (e.g., pH). Please contact us for solvent-specific guidance. |
| Purity | Purity information is available upon request (COA). |
| Synonym | Rivastigmine tartrate; 129101-54-8; Rivastigmine Hydrogen Tartrate; Rivastigmine bitartrate; ENA 713 |
| IUPAC/Chemical Name | (2R,3R)-2,3-dihydroxybutanedioic acid;[3-[(1S)-1-(dimethylamino)ethyl]phenyl] N-ethyl-N-methylcarbamate |
| InChl Key | GWHQHAUAXRMMOT-MBANBULQSA-N |
| InChl Code | InChI=1S/C14H22N2O2.C4H6O6/c1-6-16(5)14(17)18-13-9-7-8-12(10-13)11(2)15(3)4;5-1(3(7)8)2(6)4(9)10/h7-11H,6H2,1-5H3;1-2,5-6H,(H,7,8)(H,9,10)/t11-;1-,2-/m01/s1 |
| References |
3D Conformer.
(Click, turn or enlarge)
Download our GMP API Product List.
MedicaPharma is an EU-based supplier of GMP-certified APIs that serves leading healthcare institutions and research organizations.
Click here to download our full API product list.